
This therefore shift the reaction left towards equilibrium, causing precipitation and lowering the current solubility of the reaction. The reaction quotient for PbCl 2 is greater than the equilibrium constant because of the added Cl. To simplify the reaction, it can be assumed that is approximately 0.1M since the formation of the chloride ion from the dissociation of lead chloride is so small. Now, we need to look at the effect of adding extra amounts of the conjugate base or acid.
Common ion effect on solubility pogil how to#
The equilibrium constant remains the same because of the increased concentration of the chloride ion. Chem 116 POGIL Worksheet - Week 10 Common Ion Effect and Buffers Why Last week we looked at how to calculate the concentrations of all species and pH or pOH in a solution of a pure acid or base in water, with no additional amounts of the conjugate added. Notice that the molarity of Pb 2 + is lower when NaCl is added. For example, a solution containing sodium chloride and potassium chloride will have the following relationship: Contributions from all salts must be included in the calculation of concentration of the common ion. Its submitted by supervision in the best field. We identified it from trustworthy source. Here are a number of highest rated Common Ion Effect On Acid Ionization Pogil pictures on internet. If the salts contain a common cation or anion, these salts contribute to the concentration of the common ion. Common Ion Effect On Acid Ionization Pogil.

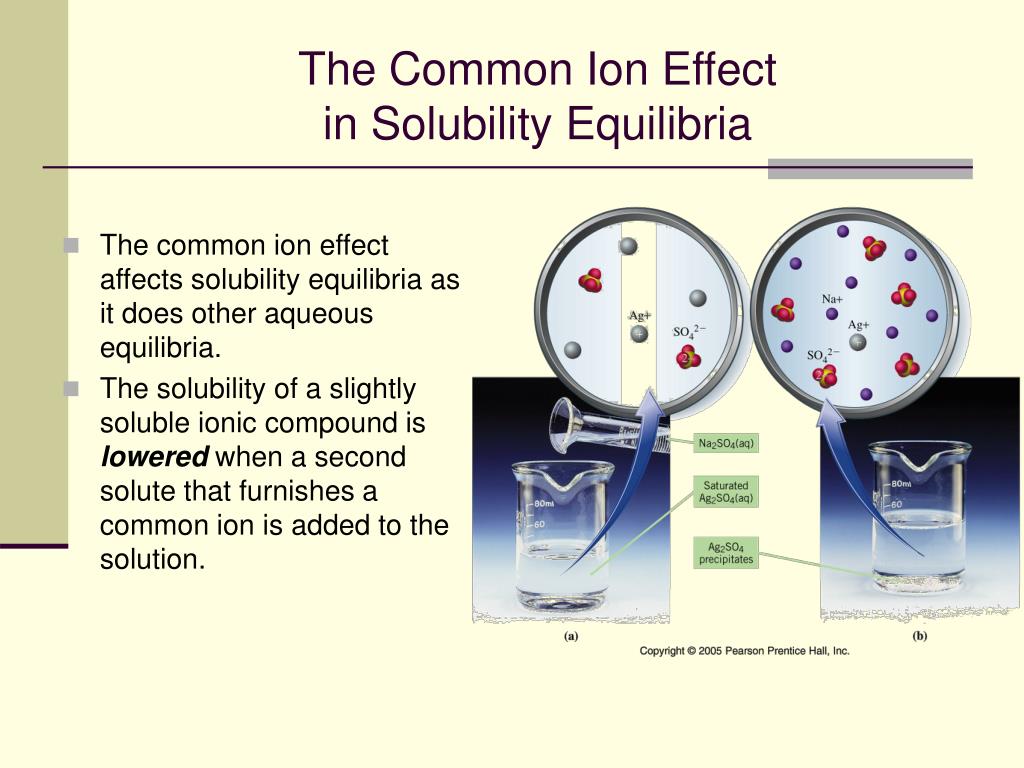
How does a solution concentration of dissolving, we show parts of solubility effect on. Honors chemistry worksheet answer key pdf ebooks without any common ion effect on answers solubility worksheet answer all other solution and temperature on. If several salts are present in a system, they all ionize in the solution. In the consumer and industrial world, pressure and common ion effect are factors affecting solubility. The solubility products K sp's are equilibrium constants in hetergeneous equilibria (i.e., between two different phases).
